-
平台编号:bio-68177
-
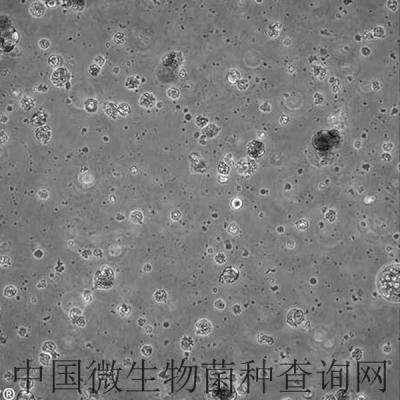
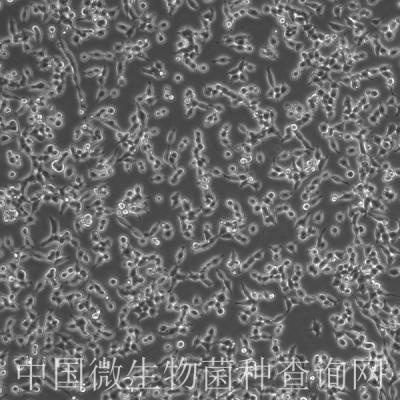
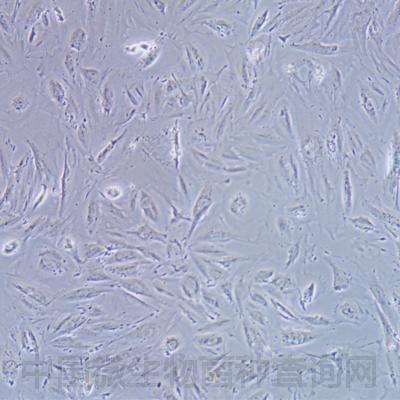
细胞信息: HFF-1
-
规格:frozen
-
用途:ATCC原装细胞
- 订购
注意事项:仅用于科学研究或者工业应用等非医疗目的不可用于人类或动物的临床诊断或治疗,非药用,非食用(产品信息以出库为准)
HFF-1
运输方式:冻存运输
细胞类型:成纤维细胞
是否是肿瘤细胞:0
物种来源:人
生长状态:贴壁生长
ATCC Number:SCRC-1041?
器官来源:皮肤
相关疾病:正常
数量:大量
细胞形态:成纤维样
年限:newborn
规格:48T Designations: HFF-1
Depositors: ?ATCC
Biosafety Level:1
Shipped: frozen
Medium & Serum: See Propagation Growth Properties:adherent
Organism: Homo sapiens
Morphology:fibroblast
Source: Organ: skin; foreskin
Disease: normal
Cell Type: fibroblast
Permits/Forms:In addition to the MTA mentioned above, other ATCC and/or regulatory permits may be required for the transfer of this ATCC material. Anyone purchasing ATCC material is ultimately responsible for obtaining the permits. Please click here for information regarding the specific requirements for shipment to your location.
Isolation: Isolation date: 2003
Applications:can be used to produce feeder cells
Age: newborn
Gender: male
Comments:The cell line was established by ATCC in 2003 from normal human foreskin pooled from two individuals. The cells can be used as a feeder layer to support the growth of embryonic stem (ES) cells and for the maintenance of ES cells in the undifferentiated state. The growth of these cells should be arrested before being used as a feeder layer. ATCC has successfully irradiated (SCRC-1041.1) and treated the cells with Mitomycin C (SCRC-1041.2) for use as a feeder layer. If the HFFs are being used as a feeder layer for ES cells, it is not recommended to use them past passage no. 50 (P50).
Propagation: ATCC complete growth medium: The base medium for this cell line is ATCC-formulated Dulbecco's Modified Eagle's Medium, Catalog No. 30-2002. To make the complete growth medium, add the following components to the base medium:
- fetal bovine serum to a final concentration of 15%
Atmosphere: air, 95%; carbon dioxide (CO2), 5%
Temperature: 37.0℃
Subculturing: Protocol: Establishing and maintaining your culture:
To insure the highest level of viability, be sure to warm media to 37?C before using it on the cells.
- Thaw the vial by gentle agitation in a 37?C water bath. To reduce the possibility of contamination, keep the O-ring and cap out of the water.
- Remove the vial from the water bath as soon as the contents are half way thawed (approximately 90 seconds), and decontaminate by dipping in or spraying with 70% ethanol. All of the operations from this point on should be carried out under strict aseptic conditions.
- Transfer the vial s contents plus 5 ml of complete medium to a 15 ml centrifuge tube. Use an additional 1 ml of medium to rinse the vial and transfer the liquid to the 15 ml tube. Add 4 ml of complete medium to bring the total volume to 10 ml.
- Gently mix and pellet the cells by centrifugation @ 270 xg for 5 minutes.
- Discard the supernatant and resuspend the cells with 10ml fresh growth medium (warm) and transfer to one coated T225 flask.
- Add 35 ml more fresh growth medium (warm) to flask.
- Incubate 37?C in a 5% CO2 in air atmosphere.
- Fluid change twice a week or when pH decreases. It is important to avoid excessive alkalinity of the medium during recovery of the cells. It is suggested that, prior to the addition of the vial contents, the culture vessel containing the growth medium be placed into the incubator for at least 15 minutes to allow the medium to reach its normal pH (7.0 to 7.6).
Subculturing Procedure:
To insure the highest level of viability, be sure to warm media and Trypsin/ EDTA to 37?C before using it on the cells.
Cells should be split when they reach confluency. A split ratio of 1:5 to 1:7 is recommended. Volumes used in this protocol are for 225cm2 (T225); proportionally reduce or increase amount of dissociation medium for culture flasks of other sizes.
- Remove and discard culture medium.
- Briefly rinse the cell layer with 1X PBS (SCRR-2201) solution to remove all traces of serum, which contain trypsin inhibitor.
- Add 5 ml of Trypsin-EDTA (0.25% (w/v) Trypsin-0.53 mM EDTA solution, ATCC # 30-2101) solution to flask and incubate for 1 minute, gently tapping the flask observe cells under an inverted microscope until cells detach (usually within 1 to 2 minutes).
- Add 6.0 to 8.0 ml of complete growth medium and rinse surface of the flask to detach all cells. Gently pipetting up and down will break cell clumps.
- Transfer all cells into a centrifuge bottle or tube and centrifuge at 270 xg for 5 minutes.
- Remove and discard the supernatant
- Add 10 ml complete growth medium to cell pellet and with 10 ml pipette resuspend the cells gently (create a single-cell suspension).
- Add more complete growth medium to cell suspension as needed to plate cells at approximately 5x10(6)/T225 flask.
- Place flasks in incubator @ 37?C with a 5% CO2 in air atmosphere.
Subcultivation Ratio: A subcultivation ratio of 1:5 to 1:7 is recommended
Medium Renewal: Twice a week or as pH decreases
Preservation: Freeze medium: Complete growth medium supplemented with an additional 40% FBS and 10% DMSO
Storage temperature: liquid nitrogen vapor phase
Related Products:Recommended medium (without the additional supplements or serum described under ATCC Medium):ATCC 30-2002
recommended serum:ATCC 30-2020
derivative:ATCC SCRC-1041.2
derivative:ATCC SCRC-1041.1
References: 89580: Amit M, et al. Human feeder layers for human embryonic stem cells. Biol. Reprod. 68: 2150-2156, 2003. PubMed: 12606388
89581: Hovatta O, et al. A culture system using human foreskin fibroblasts as feeder cells allows production of human embryonic stem cells. Hum. Reprod. 18: 1404-1408, 2003. PubMed: 12832363
89586: Andrews P, et al. Human embryonic fibroblast feeder cells. International Patent Application WO 03/078611 A1
固定电话:010-53515223
业务手机:18610886853,18610241664,18701099593,18971386853,18701098095
微信号码:vrmte521(18610886853企业微信);18610241664;biobw0905(18701098095)
企业公众号:800185668
官方邮箱:biobw11@sina.com(微生物菌种查询网)
投诉电话:biobw8888(18701099593)